Chemistry - Aromatic Hydrocarbon Question with Solution | TestHub
The Lewis acid usually reacts with the acyl halide to form an acylium ion. The acylium ion is stabilized by resonance. The acylium ion acts as an electrophile attacking the benzene ring to form an arenium ion. The arenium ion then loses a proton to generate the final product. Powerful electron withdrawing groups on the benzene ring such as another acyl group will retard this reaction. Naphthalene is the simplest and most important of the fused ring hydrocarbons. Five percent of all constituents of coal tar are naphthalene. Naphthalene can be manufactured using the Friedel-Crafts reaction via the reaction pathway shown below:
What are the most likely products of the following reaction?
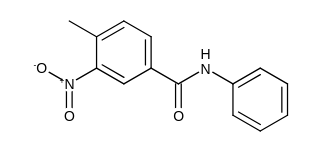
Options:
Answer:
Solution:
The reaction is a Friedel-Crafts acylation. Naphthalene undergoes electrophilic substitution preferentially at the -position (C1) due to greater resonance stabilization of the intermediate.
The correct option is B.