CHEM_REMOVED - CHEMICAL BONDING Question with Solution | TestHub
CHEM_REMOVEDCHEMICAL BONDINGVSEPR, Bond Angle, Bond Length, Bond EnergyEasy2 minPYQ_2025
CHEM_REMOVEDEasysingle choice
Given below are two statements:
Statement (I) : for , all three possible structures may be drawn as follows.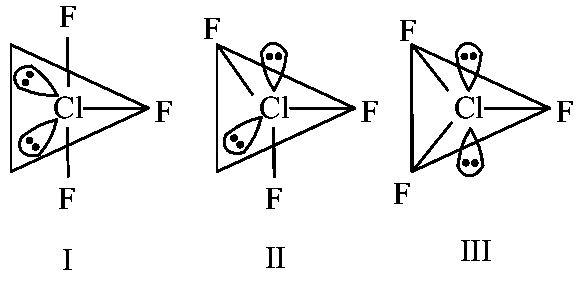
Statement (II) : Structure III is most stable, as the orbitals having the lone pairs are axial, where the repulsion is minimum.
In the light of the above statements, choose the most appropriate answer from the options given below:

Options:
Answer:
B
Solution:
Statement 1 is correct.
Statement 2 is incorrect since in hybridization; lone pair cannot occupy axial position.
Stream:JEESubject:CHEM_REMOVEDTopic:CHEMICAL BONDINGSubtopic:VSEPR, Bond Angle, Bond Length, Bond Energy
⏱ 2mℹ️ Source: PYQ_2025
Doubts & Discussion
Loading discussions...