Physics - Modern Physics Question with Solution | TestHub
PhysicsModern PhysicsAtomic structureEasy2 minPYQ_2013
PhysicsEasymultiple choice
The radius of the orbit of an electron in a Hydrogen-like atom is 4.5 a0, where a0is the Bohr radius. Its orbital angular momentum isIt is given that h is Planck's constant and R is Rydberg constant. the possible wavelength (s), when the atom de-excites, is (are)
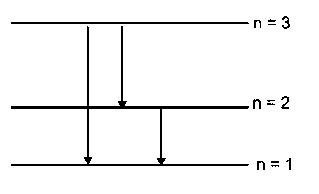
Options:(select one or more)
Answer:
A, C
Solution:

Stream:JEE_ADVSubject:PhysicsTopic:Modern PhysicsSubtopic:Atomic structure
⏱ 2mℹ️ Source: PYQ_2013
Doubts & Discussion
Loading discussions...