Physics - Heat & Thermo Question with Solution | TestHub
PhysicsHeat & ThermoFirst lawHard2 minPYQ_2021
PhysicsHardsingle choice
If one mole of an ideal gas at is allowed to expand reversibly and isothermally ( to ) its pressure is reduced to one-half of the original pressure (see figure). This is followed by a constant volume cooling till its pressure is reduced to one-fourth of the initial value Then it is restored to its initial state by a reversible adiabatic compression ( to ). The net workdone by the gas is equal to:
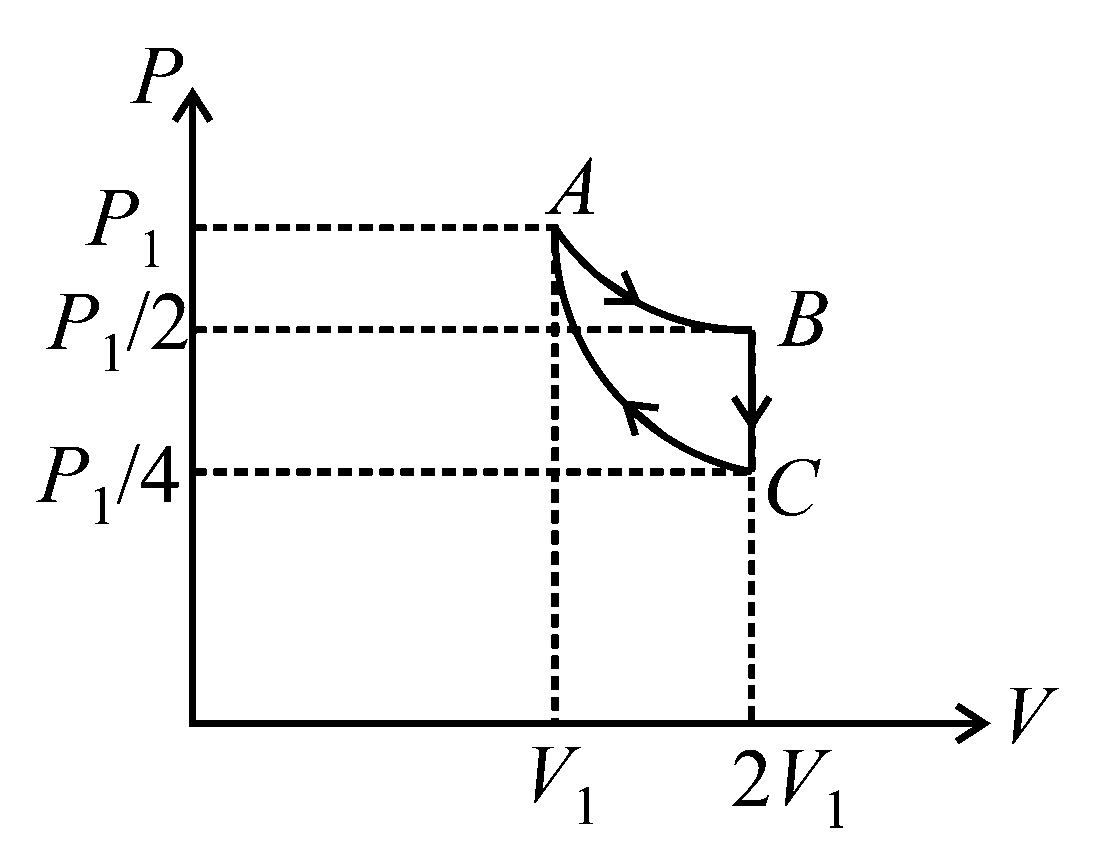

Options:
Answer:
D
Solution:
isothermal process
Isochoric process
Adiabatic process
Stream:JEESubject:PhysicsTopic:Heat & ThermoSubtopic:First law
⏱ 2mℹ️ Source: PYQ_2021
Doubts & Discussion
Loading discussions...