Physics - Heat & Thermo Question with Solution | TestHub
PhysicsHeat & ThermoDifferent processesEasy2 minPYQ_2014
PhysicsEasysingle choice
One mole of diatomic ideal gas undergoes a cyclic process ABC as shown in figure. The process BC is adiabatic. The temperatures at A, B and C are 400 K, 800 K and 600 K respectively. Choose the correct statement :
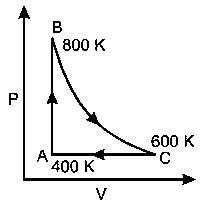

Options:
Answer:
D
Solution:
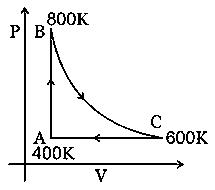
Stream:JEESubject:PhysicsTopic:Heat & ThermoSubtopic:Different processes
⏱ 2mℹ️ Source: PYQ_2014
Doubts & Discussion
Loading discussions...