CHEM_REMOVED - CHEMICAL BONDING Question with Solution | TestHub
CHEM_REMOVEDCHEMICAL BONDINGIntroduction, ValencyEasy2 minPYQ_2022
CHEM_REMOVEDEasysingle choice
Which of the following pair of molecules contain odd electron molecule and an expanded octet molecule?
Options:
Answer:
B
Solution:
An odd electron bond means there is an odd number of the electron in the overall molecule. Or we can say that the molecule has unpaired electrons.
So, in , the valence electrons in nitrogen are , and valence electrons in oxygen are . So, by adding we get,
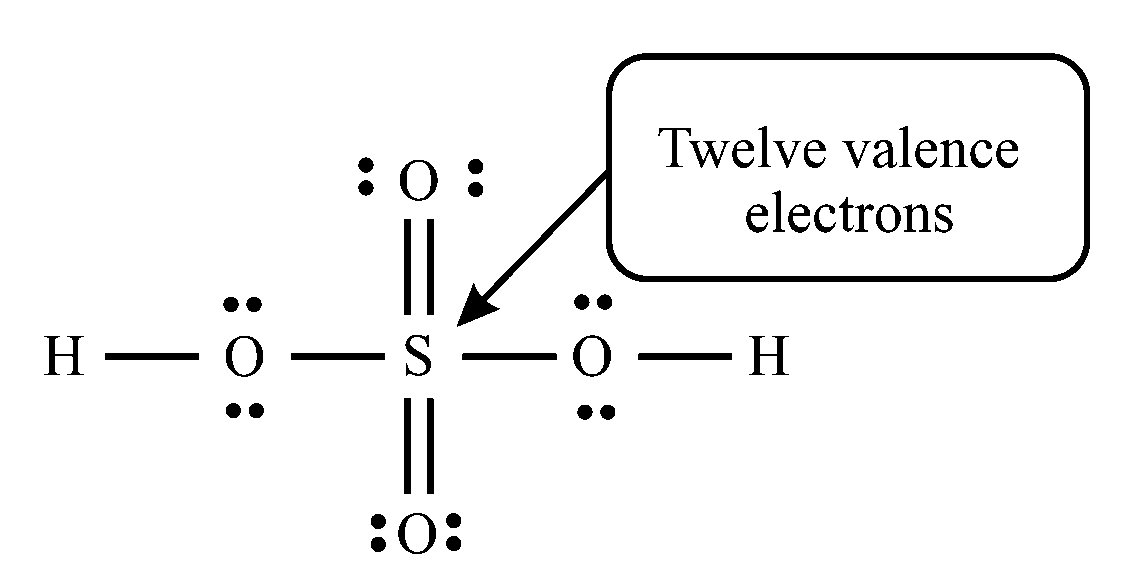
In sulfuric acid , each oxygen has a full octet (eight valence electrons), whereas sulfur has an expanded octet (twelve valence electrons).
(A) Even Electron molecule Expanded octet molecule
(B) Odd Electron molecule Expanded octet.
(C) Even Electron molecule Expanded octet.
(D) Even Electron molecule Odd Electron molecule
Stream:JEESubject:CHEM_REMOVEDTopic:CHEMICAL BONDINGSubtopic:Introduction, Valency
⏱ 2mℹ️ Source: PYQ_2022
Doubts & Discussion
Loading discussions...