CHEM_REMOVED - CHEMICAL BONDING Question with Solution | TestHub
CHEM_REMOVEDCHEMICAL BONDINGVSEPR, Bond Angle, Bond Length, Bond EnergyMedium2 minPYQ_2018
CHEM_REMOVEDMediumsingle choice
Which of the following conversions involves change in both shape and hybridisation?
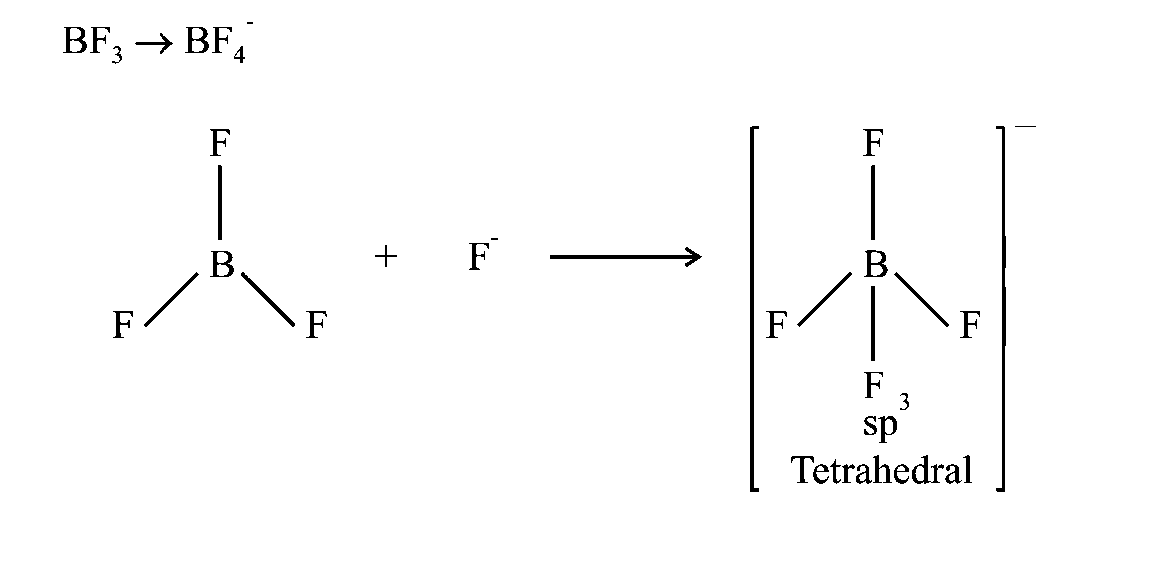
Options:
Answer:
B
Solution:
Hybridization in Chemistry is defined as the concept of mixing two atomic orbitals to give rise to a new type of hybridized orbitals. This intermixing usually results in the formation of hybrid orbitals having entirely different energies, shapes, etc. The atomic orbitals of the same energy level mainly take part in hybridization. However, both fully filled and half-filled orbitals can also take part in this process, provided they have equal energy.
Here, has hybridisation while has hybridisation.

Stream:JEESubject:CHEM_REMOVEDTopic:CHEMICAL BONDINGSubtopic:VSEPR, Bond Angle, Bond Length, Bond Energy
⏱ 2mℹ️ Source: PYQ_2018
Doubts & Discussion
Loading discussions...